Like it or not, every major auto manufacturer is working with hydrogen fuel cells - and there's a reason for that. Hydrogen is extremely energy dense, available everywhere in the world (or universe, for that matter), and can be used in place of heavier, less energy dense batteries in automotive applications for electric vehicles.
With all of that, hydrogen has some down-sides. Daihatsu thinks it has found a solution to eliminate those downers and make fuel cells cheaper and their fuel more safe in storage and transport. They showed off their ideas in Tokyo with their FC ShoCase concept vehicle.
The ShoCase itself is a pure concept that will not ever likely make it to production. It was shown as part of the company's Kei line of ultra-small vehicles for urban use. These vehicles sell well in Japan and Daihatsu, as the country's oldest automaker, is a small, but innovative and proud company. Since 2007, they've been working with an alternative fuel concept for fuel cell vehicles.
While the rest of the world's carmakers, including Daihatsu's part-owner Toyota, are perfecting hydrogen fuel cells, Daihatsu is instead working with hydrazine.
If you're familiar with hydrazine, it's likely because you're familiar with polymer foams, rocket fuels, or the manufacture of air bags or pharmaceuticals. Or perhaps with nuclear power plant operations. This chemical is used in dozens of applications for which some 260,000 tons are produced annually. In its purest form, it's extremely toxic, but it's most often used in solution. Hydrazine is comparable to ammonia with a chemical signature of N2H4.
So why use it in place of hydrogen for fuel cells? Simple. Everything bad about fuel cells becomes good with hydrazine.
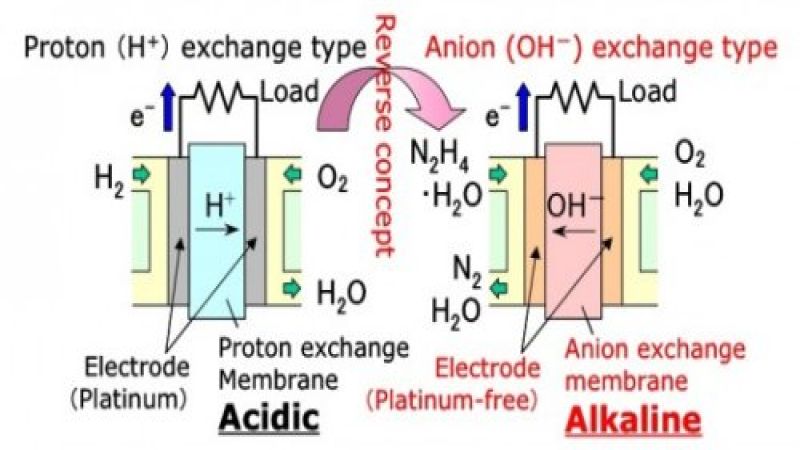
Hydrogen's biggest dilemma in fuel cell use is its requirement that it be bathed in highly acidic electrolyte solution in order to facilitate the proton exchanges necessary to give off that coveted electron to run through an electrical circuit. This acidic chemistry means that most metals will not last long and thus cannot be used as a catalyst. Top choices are narrow and usually expensive, with most using platinum as their choice. This is what makes fuel cells expensive, potentially dangerous if ruptured, and relatively hard to build. Add to that the highly volatile nature of hydrogen itself, one of the most flammable gases known, and there are some obvious obstacles to overcome with hydrogen fuel cells.
Hydrazine, on the other hand, has none of those problems. Hydrazine works best in a fuel cell that is aklaline, rather than acidic, and so just about any cheap, readily available metal can be used as a catalyst. Daihatsu has tried both cobalt and nickel with success. What's more, if the fuel cell is ruptured, nothing inside need be toxic to the environment or people, making it much safer.
But wait, isn't hydrazine toxic? Only if it's in its pure form. Daihatsu has instead come up with a fuel tank for storing it that fixes the hydrazine hydrate solution to the fuel tank itself, using a polymer coating, which minimizes spill potential and does not hinder fuel delivery. It should also be mentioned that hydrazine hydrate is a liquid fuel that can be distributed in the same way gasoline is today, with the same general safety precautions, but none of the flammable fume worries gasoline has. It can also be synthetically produced without petroleum and gives off no CO2 or toxins once used (it releases only water and nitrogen from the tailpipe).
So far, engineers at Daihatsu have managed to produce an output density of 0.50 watts per centimeter squared (W/cm2), which is comparable to current hydrogen fuel cells.
Interestingly, few in the automotive press have noticed this little entry from Japan's most venerable car maker. Most seem to have been instead interested only in the ShoCase concept car rather than its highly innovative workings under the hood (if it had a hood). It's possible that Daihatsu has made the breakthrough that hydrogen fuel cell developers have been pining after for years.











Comments
One of the big advantages you
Permalink
One of the big advantages you get with hydrogen gas over a liquid is safety.
Yes, contrary to popular images (Hindenburg) a gas is safer than a liquid because in a crash, the hydrogen gas quickly evaporates into the atmosphere unlike gasoline which pools to the ground and engulfs the people in flames.
I also hesitate to think why you would want to take an harmless gas like hydrogen which is perfectly useable and replace it with a toxic poison like hydrazine.
No. I don't like this idea at all...
Having handled hydrogen gas
Permalink
In reply to One of the big advantages you by John Bailo (not verified)
Having handled hydrogen gas personally and having held a Hazardous Materials license to haul it, I can say that your impressions of hydrogen gas are largely incorrect. It's NOT safer to transport or pump. In fact, it's the least safe of the flammable gases to handle. Especially under pressure. In a crash, it doesn't "dissipate into the air" it EXPLODES into the air in a fireball.
Did I mention that hydrazine hydrate doesn't require expensive compressors or handling equipment either?
Sorry, but I can't believe
Permalink
In reply to Having handled hydrogen gas by Aaron Turpen
Sorry, but I can't believe you one bit....
Then I would suggest that you
Permalink
In reply to Sorry, but I can't believe by John Bailo (not verified)
Then I would suggest that you educate yourself. I've listed my credentials and reasoning, so there's nothing more I can give you.
What? Hydrazine? I remember
Permalink
What? Hydrazine? I remember when they used hydrazine in dragters many, many years back. Yes, explosions. Talk about an unstable fuel!!!!
This is hydrazine hydrate,
Permalink
In reply to What? Hydrazine? I remember by Frank Sherosky
This is hydrazine hydrate, which is not explosive. Very different from the rocket fuel hydrazine you're talking about.
The whole plan is
Permalink
In reply to This is hydrazine hydrate, by Aaron Turpen
The whole plan is insane.
Even compressed hydrogen is a known technology.
KIA is rolling out production model hydrogen fuel cell cars for Europe right now...this year!
Plus hydrogen can be generated by anyone from sunlight, wind and dam power.
Changing a perfectly safe gas technology into a poisonous liquid borders at the edge of mental illness.
They aren't in production,
Permalink
In reply to The whole plan is by John Bailo (not verified)
They aren't in production, only building prototypes. They plan to enter production in 2014-15.
Hydrazine can be produced in the same way hydrogen is produced. The nitrogen is all around us, all the time. It's a simple chemical process. Neither hydrogen nor hydrazine is produced from electrolysis, though - most comes from natural gas. And it will continue to be so for the foreseeable future because the other methods are not sustainable in production quantity.
Again, I pointed out the problems with hydrogen fuel cells in this article - problems which you've completely ignored in favor of claiming that H2 is "safe" - which it isn't.
You're bordering on religious zealotry here. This is an alternative that is cheaper to use, just as cheap to get, and that can go into production (in terms of automotive) much faster and at lower consumer costs than hydrogen fuel cells can.
US Army Labs at Fort Belvoir,
Permalink
US Army Labs at Fort Belvoir, VA demonstated hydrazine fuel cells in the 1960's. A 300 watt units was field tested in Viet Nam and a 20 k/W unit powered a military type pick up truck. The goal always was to use logistic fuels which were clearly far into the future. Hydrazine hydrate fuel cells allowed testing of systems using electric power source to provide a basis for hydrocarbon fuel cell research.
my 1999 lincoln town car air
Permalink
my 1999 lincoln town car air suspension is not holding the
It sometimes felt like I couldn't go anywhere without being bothered. I was "recognized" at church, at the gym, by one of the clients at Haven for Hope. My fellow journalists here at the Express News never seem to tire of needling me whenever my clone does something newsworthy like leaving the White House and being elected mayor of Chicago..
The ignition switch on the steering column of the Chevrolet Cobalt and other small cars was so poorly designed that it easily slipped out of the run position, causing engines to stall. Engineers knew it; as early as 2004, a Cobalt stalled on a GM test track when the driver's knee grazed the key fob. By GM's admission, the defective switches caused over 50 crashes and at least 13 deaths..
How can we help it?" LIAM BARTLETT: If you're out there doing all that amongs the plastic and the rubbish it sort of takes the gloss off it. LAIRD HAMILTON: The ocean full of plastic is an ugly place. IAN KIERNAN: What you've got to realise whatever you drop on a mountain or in a creek or on the beach or in the street or in the paddock is eventually most likely to end up in the ocean, the ultimate receiving waters.
?>
Promising article: thank you,
Permalink
Promising article: thank you, Any updates?
Also, re hydrogen as a fuel, I believe that is a difficult gas to compress, & depending on the required operating pressure, , one ends up using about ~30% of its specific energy getting the H2 to useful pressure. Of course it’s specific energy density is very high. But still that’s a ~30% efficiency hit right there.