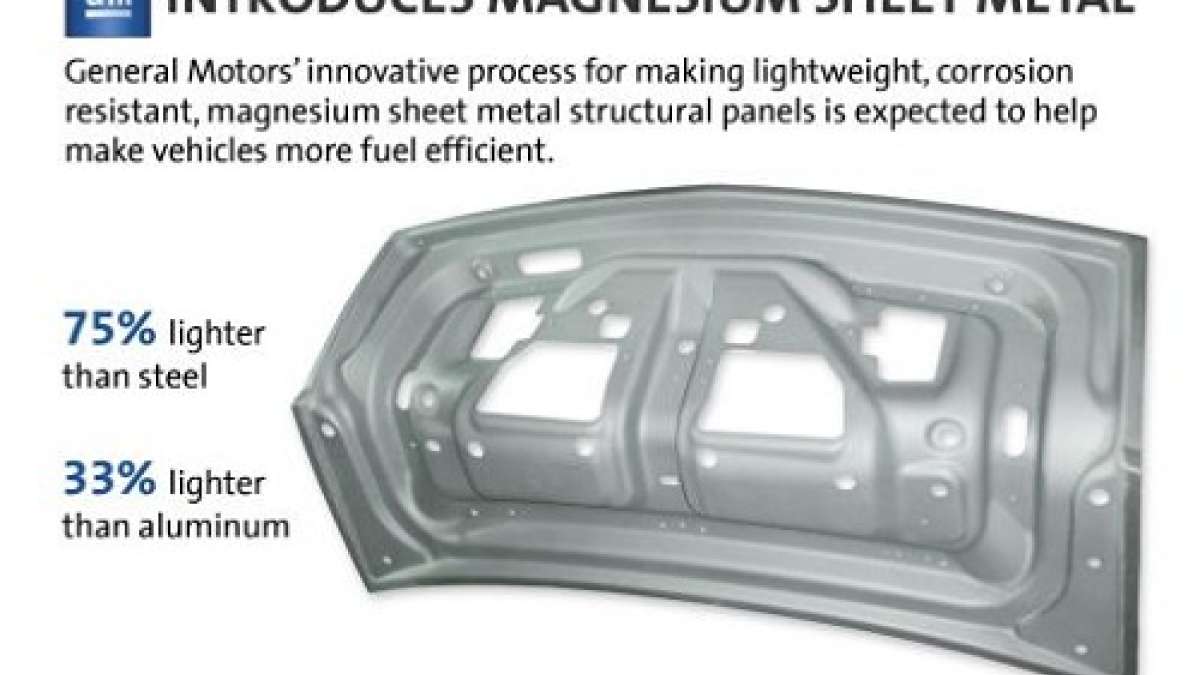
In the quest to add lightness to automobiles GM has always been a pioneer. Few automakers have done as much with Sheet-molded-composite, sometimes called fiberglass, as has General Motors in the Chevrolet Corvette. Aluminum is no stranger to GM products as well. Even titanium will occasionally be tapped for use in a GM product when its cost benefit analysis comes out the right way. One material has properties that rival these low-weight materials and is a metal that is both rigid and strong. It is 33% lighter than aluminum, 60% lighter than titanium and much easier to work than titanium. That material is magnesium.
Magnesium has been used in automobiles for about 100 years. It has had particular notoriety in racing cars. It entered service as part of a mainstream production car at VW in the pre-WWII era, and is stull used in cast parts in many cars. However, it has never been used widely (or at all) in a production car in sheet metal form. General Motors has now engineered a process to make magnesium into sheets that can then be stamp-formed into complex flat shapes. GM’s trick is to heat the metal to 842 degrees Fahrenheit before metal forming it in to very exact shapes.
It is ironic that GM uses heat to solve this riddle because magnesium’s past includes some real horror stories involving fire. Magnesium can auto ignite at temperatures not much higher than the heat-forming temperature GM is using in its production. Magnesium does not burn easily in bulk, but shavings burn too well. In fact, most firefighting methods do not extinguish burning magnesium. Water and CO2 are not helpful in putting out fires involving magnesium.
Another issue involving magnesium is corrosion. Although it forms an oxide barrier like aluminum, scratches can result in corrosion. The material is also not fond of acids or salts, both of which are found in the water cars come in contact with. GM has found a proprietary way to passivate magnesium that will withstand salt spray. Used in a deck-lid as the inner structure it is also able to withstand the rigors of trunk slams. General Motors R&D metals researcher Jon Carter was quoted in a press release as saying “Like all of our advanced material vehicle parts, we subjected the magnesium trunk lid to the most severe strength and corrosion tests we know of, and it passed with flying colors. We expect it to perform extremely well even in the harshest environments.”
GM says its goal will be to ultimately save about 300 pounds by substituting magnesium where steel and aluminum are now used. It claims that a fuel savings of from 9 to 12 percent can be achieved if this goal is met.











Comments
Please forgive my jab: "Add
Permalink
Please forgive my jab:
"Add lightness?" So does that mean that it could be possible that the new trunk lid could be "twice as light?"
I have always believed that magnesium is immensely expensive. Does this GM development force me to toss away another long-held "truth" from my past?
I truly enjoy reading about these significant but not very sexy details about the auto industry. Keep it up!